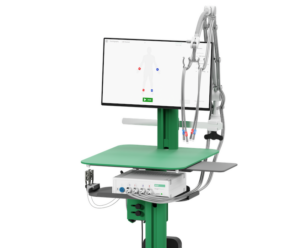
AngE™ Phlebo
Venous and Arterial PPG Sensor
🇦🇹 Made in Austria
AngE Phlebo Venous - EN – 1.74 MB
Request Now2-Channel D-PPG/LRR
Simultaneous Venous Function Tests to assess Venous Valve Incompetence and Muscle Pump function.
Request NowVenous Function Measurements
The AngE Phlebo is a 2-channel D-PPG/LRR device. This means it uses two IR-sensors to perform a venous function measurement. The system also supports using a set of Tourniquet cuffs.
Pump Volume and Fill Time
AngE Phlebo calculates pump volume (V0), venous fill time (T0) and venous half-life time (T50) automatically. The results are stated in a simple traffic light display and allow for a fast venous valve incompetence diagnostics.
Manual Tourniquet Option
Apply manual tourniquet cuffs to rapidly differentiate between superficial and deep venous problems.
Muscle Pump Function
In order to evaluate the function of the muscle pump, AngE Phlebo allows performing measurements while the patient is walking.
Temperature Probes
Measurement probes on both sensors sense and compare the patient's skin temperature during the measurement.
Comprehensive Software
AngE Phlebo comes with a sophisticated macOS and Windows software featuring patient management, measurement analysis, DICOM/HL7 interfaces and many more.
Venous Function Tests
For a fast assessment of venous valve incompetence.
Determine pump volume (V0), venous fill time (T0) and venous half-life time (T50) in one simple test.
Test Protocols
Included or extendable measurement methods.
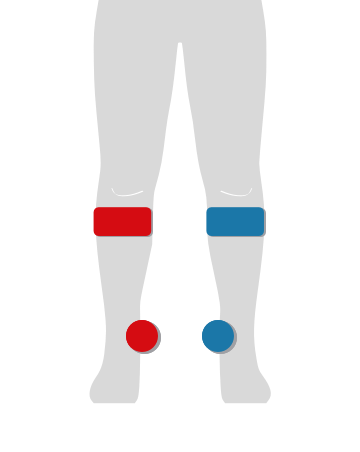
CVI
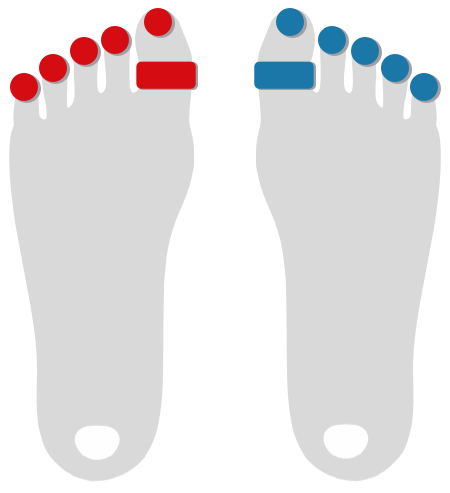
Toe Pressure
Raynaud's Syndrome

TOS

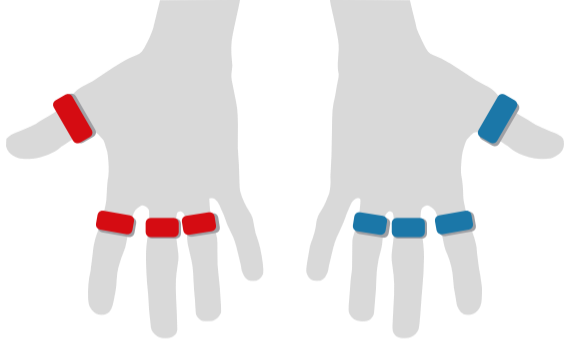
Allen's Test
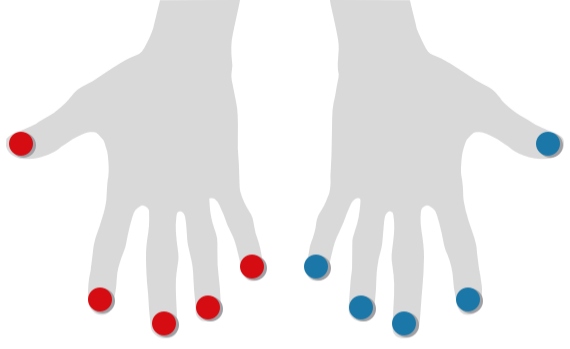
Finger Flow
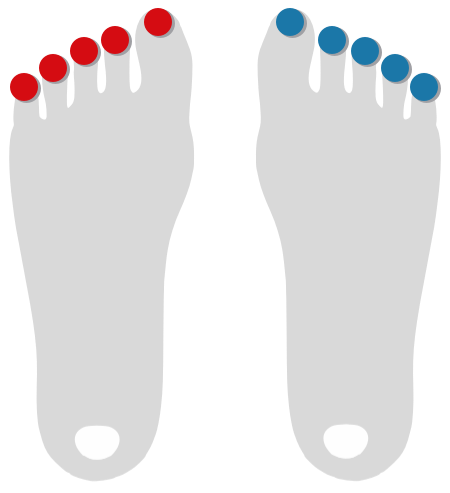
Toe Flow

Add Custom Tests
ABI, TBI, Toe Pressure and PWV
Multiple arterial and venous parameters within one mobile system.
Temperature Probes
Measurement probes on both sensors sense and compare the patient's skin temperature during the measurement.
Mobile USB-Powered Device
Simply plug in to the USB port of a macOS or Windows computer, open the easy-to-use software and start the test.
DICOM / HL7 Compatible
AngE Phlebo comes with a sophisticated software featuring patient management, measurement analysis, DICOM/HL7 interfaces and much more.
Exercise Stress-Tests
Use the build-in comparison tool for Exercise Stress-Test to detect differences pre and post provocation.
Comparison Pre- and Post Intervention
The easy to use software allows for direct comparisons pre- and post-intervention.
Optical Pulse Oscillography on Fingers and Toes
Assess Microcirculation for effective evaluation of arterial circulatory disorders, such as Raynaud's Syndrome or TOS.
Photoplethysmography (PPG) with Optical Pulse Oscillography (OPO) allows to determine the patient's arterial microcirculatory blood flow in toes and fingers.
It therefore makes rapid examinations of TOS (Thoracic Outlet Syndrome) as well as the conduction of Toe Pressure measurements, Raynaud Syndrome studies, and more, possible.
The AngE Phlebo is the state-of-art, haemodynamically significant D-PPG system for venous diagnostics. This non-invasive functional investigation has always helped me accurately examine venous disorders, even with complex cases.
The high sensitivity of the optical sensors allow for a good documentation of the pulse waves, even with marginal blood flow. Given the virtually unfiltered display of pulse curves, dicrotic waves can be clearly identified for healthy and elastic arteries.
What's in the Box:

AngE Phlebo PPG + Software*
Adhesive Pads

Manometer

Cuffs

Backpack
Extendable with AngE™ ABI+
Combine AngE™ Phlebo with the AngE™ ABI+ to do automated ABI, Toe Pressure and TBI tests.
AngE™ ABI+
AngE™ Phlebo

AngE™ ABI+
Get a Quote
Your local representative will be in contact shortly.
* AngE Phlebo is a 2-probe infrared device powered by its USB port. A computer with available USB port, operated with Windows 11 or mac OS is required for operation. Computer not included. More information.
CE-marked device: Our AngE products comply with the requirements of RoHS 2011/65/EC and Annex IV of the Medical Device Directive 93/42/EC. More information. Personal face-to-face training is required before operating this device.
References
1. Interrater and intrarater reliability of photoplethysmography for measuring toe blood pressure and toe- brachial index in people with diabetes mellitus. (Scanlon C. et al. 2012)
2. Photoplethysmography detection of lower limb peripheral arterial occlusive disease: a comparison of pulse timing, amplitude and shape characteristics. (Allen J et al. 2005)
3. Photoplethysmography can replace hand-held Doppler in the measurement of ankle/brachial indices. (Whitely MS et al. 1998)
4. Effects of isokinetic calf muscle exercise program on muscle strength and venous function in patients with chronic venous insufficiency. (Ercan et al. 2018)
5. How posture influences venous blood flow in the lower limbs: results of a study using photoplethysmography. (Lastowiecka-Moras 2017)
6. An evaluation of the lower extremity muscle strength of patients with chronic venous insufficiency. (Cetin et al. 2016)
7. Risk factors for chronic ulceration in patients with varicose veins: a case control study. (Robertson et al. 2009)
Instant Support
in one minute
Our product experts in Austria will help you with remote support without any waiting time or ticket system. Call us during office hours and we will help you within a minute.
Mo - Th: 8am - 5pm, Fr: 8am - 12am